4759. Hycanthone
Nomenclature
CAS number: 3105-97-3
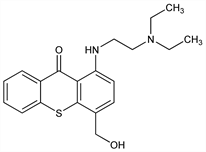
1-[[2-(Diethylamino)ethyl]amino]-4-(hydroxymethyl)-9H-thioxanthen-9-one. C20H24N2O2S; mol wt 356.48.
C 67.38%, H 6.79%, N 7.86%, O 8.98%, S 8.99%.
Description and references
Metabolite of lucanthone, q.v.: Rosi et al., Nature 208, 1005 (1965). Prepn by oxidative fermentation of lucanthone and schistosomicidal activity: Rosi et al., J. Med. Chem. 10, 867 (1967); NL 6410359, and Rosi, Peruzzotti, US 3294803; US 3312598 (1965, 1966, 1967 all to Sterling Drug). Alternate synthesis: Laidlaw et al., J. Org. Chem. 38, 1743 (1973).
Properties
Crystals, mp 100.6-102.8°. Absorption max (ethanol): 233, 258, 329, 438 nm (ε 19400, 37000, 9700, 6600). Extremely sensitive to acid.Derivative
Hydrochloride.Properties
mp 173-176° (dec).Derivative
Mesylate.Nomenclature
CAS number: 23255-93-8
Etrenol (Winthrop). Therapeutic Category
Anthelmintic (Schistosoma).
Keywords
Anthelmintic (Schistosoma)