4763. Hydralazine
Nomenclature
CAS number: 86-54-4
1(2H)-Phthalazinone hydrazone; 1-hydrazinophthalazine; Ciba 5968; Prparat 5968; C-5968. C8H8N4; mol wt 160.18.
C 59.99%, H 5.03%, N 34.98%.
Description and references
Prepd by the action of hydrazine hydrate on 1-chloro- or 1-phenoxyphthalazine: Hartmann, Druey, US 2484029 (1949 to Ciba); Druey, Ringier, Helv. Chim. Acta 34, 204 (1951). Metabolism: Z. H. Israile, P. G. Dayton, Drug Metab. Rev. 6, 283 (1977); K. Schmid et al., Arzneim.-Forsch. 31, 1143 (1981). Pharmacology: J. L. Cangiano et al., J. Lab. Clin. Med. 92, 516 (1978). Clinical paper: R. F. Albrecht et al., Int. Anesthesiol. Clin. 16, 299 (1978). Acute toxicity: L. Dorigotti et al., Pharmacol. Res. Commun. 8, 295 (1976). Comprehensive description: C. E. Orzech et al., Anal. Profiles Drug Subs. 8, 283-314 (1979).
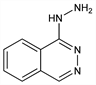
Properties
Yellow needles from methanol, mp 172-173° (rapid heating). One gram dissolves in 3 ml 2N acetic acid, in 12 ml warm methanol. Forms a red compd (phthalazinylhydrazone) with acetone at 60° in presence of 2N acetic acid. LD50 in mice, rats (mg/kg): 122, 90 orally; 101, 40 i.p. (Dorigotti).Derivative
Hydrochloride.Nomenclature
CAS number: 304-20-1
Alphapress (Alphapharm); Apresoline (Novartis). C8H8N4.HCl; mol wt 196.64.
C 48.86%, H 4.61%, N 28.49%, Cl 18.03%.
Properties
Yellow crystals, dec 273°. Soly in water (g/100 ml) at 15°: 3.01; at 25°: 4.42; in 95% ethanol: 0.2 g/100 ml. Very slightly sol in ether. pH of a 2% aq soln 3.5 to 4.5. uv max (0.001% aq soln): 211, 240, 260, 304, 315 nm. Aq solns containing 20 mg/ml may be preserved with 0.5% chlorobutanol.Therapeutic Category
Antihypertensive.
Keywords
Antihypertensive; Hydrazines/Phthalazines