4806. Hydroquinidine
Nomenclature
CAS number: 1435-55-8
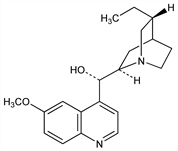
(9S)-10,11-Dihydro-6′-methoxycinchonan-9-ol; dihydroquinidine; hydroconchinine. C20H26N2O2; mol wt 326.43.
C 73.59%, H 8.03%, N 8.58%, O 9.80%.
Description and references
An alkaloid of cinchona, stereoisomeric with hydroquinine. Usually prepd by hydrogenation of quinidine: Heidelberger, Jacobs, J. Am. Chem. Soc. 41, 826 (1919). Conversion to dihydrocinchonine by removal of the methoxy group: King, J. Chem. Soc. 1946, 523. Manuf pat.: Gutzwiller, Uskokovic, DE 1933599 (1970 to Hoffmann-La Roche), C.A. 72, 90696v (1970). Pharmacology: Cosnier et al., Therapie 26, 97 (1971).
Properties
Plates from ether, needles from alcohol, mp 169°. [α]D20 +231° (c = 2.02 in alc); +299° (c = 0.82 in 0.1N H2SO4). Readily sol in hot alcohol; slightly sol in water and ether.Derivative
Hydrochloride.Nomenclature
CAS number: 1476-98-8
Serecor (Houdé). C20H26N2O2.HCl; mol wt 362.89.
C 66.19%, H 7.50%, N 7.72%, O 8.82%, Cl 9.77%.
Properties
Rhombic plates, mp 273-274°. [α]D26 +184° (c = 1.3). Freely sol in methanol, chloroform; less readily in water or abs alcohol; difficultly sol in dry acetone.Therapeutic Category
Antiarrhythmic.
Keywords
Antiarrhythmic