4810. Hydroxyamphetamine
Nomenclature
CAS number: 1518-86-1
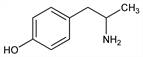
4-(2-Aminopropyl)phenol; dl-p-hydroxy-α-methylphenethylamine; dl-1-p-hydroxyphenyl-2-propylamine; p-hydroxyphenylisopropylamine; α-methyltyramine; Paredrine (SK & F); Paredrinex (SK & F); Pulsoton. C9H13NO; mol wt 151.21.
C 71.49%, H 8.67%, N 9.26%, O 10.58%.
Description and references
Prepn from oxime of p-methoxyphenyl acetone: Mannich, Jacobsohn, Ber. 43, 189 (1910), DE 243546. From p-nitrobenzyl chloride and a salt of nitroethane: Hoover, Hass, J. Org. Chem. 12, 501 (1947).
Properties
Crystals (rosettes) from benzene, mp 125-126°. Sol in water, alcohol, chloroform, ethyl acetate.Derivative
Iodide.C9H14INO; mol wt 279.12.
C 38.73%, H 5.06%, I 45.47%, N 5.02%, O 5.73%.
Properties
Stout prisms, mp 155°. Freely sol in water, alcohol, acetone.Derivative
Hydrochloride.C9H14ClNO; mol wt 187.67.
C 57.60%, H 7.52%, Cl 18.89%, N 7.46%, O 8.53%.
Properties
Crystals from HCl, mp 171-172°. Sol in water, alcohol. Practically insol in ether.Derivative
Hydrobromide.Nomenclature
CAS number: 306-21-8
C9H14BrNO; mol wt 232.12.
C 46.57%, H 6.08%, Br 34.42%, N 6.03%, O 6.89%.
Properties
Crystals. Freely sol in water, alcohol, acetone. Prepd as Paredrine Hydrobromide Aqueous—a 1% aq soln made isotonic with sodium chloride and preserved with sodium ethylmercuri thiosalicylate. Also prepd as Paredrine Hydrobromide Ophthalmic 1%, with boric acid—a 1% aq soln made tear-isotonic with 2% boric acid and preserved with sodium ethylmercuri thiosalicylate.Therapeutic Category
Adrenergic (ophthalmic); mydriatic.
Keywords
α-Adrenergic Agonist; Mydriatic