4816. β-Hydroxybutyric Acid
Nomenclature
CAS number: 300-85-6
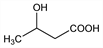
3-Hydroxybutanoic acid. C4H8O3; mol wt 104.10.
C 46.15%, H 7.75%, O 46.11%.
Description and references
Prepd from acetoacetic ester by the action of sodium amalgam: Wislicenus, Ann. 149, 207 (1869); Marian, Biochem. Z. 150, 283 (1924); by the oxidation of aldol: Wurtz, Compt. Rend. 76, 1167 (1873); from crotonic acid by heating with dil acid: Wacker, DE 441003; Frdl. 15, 135; by heating crotonitrile with KOH soln: Bruylants, Bull. Soc. Chim. Belg. 31, 182 (1922).
Properties
Hygroscopic syrup. Volatile with steam. Sol in water, alcohol, ether. On distn it dec into crotonic acid and water.Derivative
d-Form.Nomenclature
CAS number: 6168-83-8
(3S)-3-Hydroxybutanoic acid. Description and references
Prepd by the action of Aspergillus griseus on the dl-form: McKenzie, Harden, J. Chem. Soc. 83, 430 (1903).
Properties
Crystals. [α]D10 +24.3° (c = 2.226). Sol in water, alcohol, ether.Derivative
l-Form.Nomenclature
CAS number: 625-72-9
(3R)-3-Hydroxybutanoic acid. Description and references
Found in the urine of diabetics (as much as 30 g per day). Isoln: Fischer, Scheibler, Ber. 42, 1221 (1909); Shaffer, Marriott, J. Biol. Chem. 16, 268 (1913).