4845. Hydroxystilbamidine
Nomenclature
Description and references
Prepn: J. N. Ashley, J. O. Harris, J. Chem. Soc. 1946, 567; A. J. Ewins et al., GB 574486; A. J. Ewins, US 2510047 (1946, 1950 both to May & Baker). Organ and tissue distribution in animals: I. Snapper et al., Cancer 4, 1246 (1951). Pharmacology and antiprotozoal activity: I. Snapper et al., Trans. N.Y. Acad. Sci. 14, 269 (1952). Probe for studying nucleic acid conformation: B. Festy, C. R. Seances Acad. Sci. Ser. D 266, 1433 (1968); B. Festy, M. Daune, Biochemistry 12, 4827 (1973); B. Festy et al., Biochim. Biophys. Acta 407, 24 (1975). Crystal structure: C. Courseille et al., C. R. Seances Acad. Sci. Ser. C 274, 1921 (1972). Use as a fluorochrome for selective staining of nuclei: L. B. Murgatroyd, Histochemistry 74, 107 (1982). Review: B. Festy in Antibiotics vol. 5, pt. 2, F. E. Hahn, Ed. (Springer-Verlag, New York, 1979) pp 223-235.
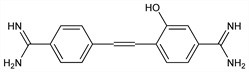
Properties
Yellow microcrystals from nitrobenzene, mp 235°. LD50 in mice (mg/g); 0.027 i.v.; 0.14 s.c. (Ewins, 1950).Derivative
Isethionate.Nomenclature
Properties
Yellow crystals, discolored by light, mp 286° (dec). Freely sol in water. Soly in alcohol ≈1.0 g/100 ml. Practically insol in ether. pH of a 1% aq soln 3.3 to 5.3. Solns show strong yellow fluorescence under uv light. Solns should be freshly prepared. Although the hydroxy compd is more stable in soln, such solns should not be stored and must not be used if cloudy.Therapeutic Category
Antiprotozoal (Leishmania).
Keywords
Antiprotozoal (Leishmania)