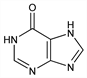
4869. Hypoxanthine
Nomenclature
Description and references
Desmotropic forms: purin-6-ol; 1H-purin-6-ol; 3H-purin-6-ol; 9H-purin-6-ol; purin-6(3H)-one; 9H-purin-6(1H)-one. Formed in the animal body during the breakdown of nucleic acids; formation from adenosine continues after death. Also widely distributed in the vegetable kingdom. Numerous syntheses, e.g., from 2,6,8-trichloropurine: Fischer, Ber. 30, 2226 (1897); by oxidation of adenine: Krüger, Z. Physiol. Chem. 18, 445 (1894). By reduction of uric acid: Sundwik, ibid. 76, 486 (1912); by condensing ethyl cyanoacetate and thiourea in the presence of Na-ethoxide: Traube, Ann. 331, 64 (1904); from mercapto-4-hydroxy-6-aminopyrimidine: Taylor, Cheng. J. Org. Chem. 25, 148 (1960). Reviews: Levene, Bass, ACS Monograph Series no. 56, entitled “Nucleic Acids” (New York, 1931); Chargaff, Davidson, Nucleic Acids vols. I & II (Academic Press, New York, 1955).