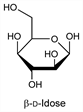
4889. Idose
Nomenclature
Description and references
Prepn of d-idose by reduction of d-idonolactone: Fischer, Fay, Ber. 28, 1975 (1895); from d-galactose: Sorking, Reichstein, Helv. Chim. Acta 28, 1 (1945); from tri-O-acetyl-1,6-anhydro-β-d-idopyranose: eidem, ibid. 662. Prepn of l-idose by reduction of l-idonolactone: Fischer, Fay, loc. cit.; by hydrolysis of 1,2-O-isopropylidene-l-idofuranose: Meyer, Reichstein, Helv. Chim. Acta 29, 152 (1946); von Vargha, Ber. 87, 1351 (1954). Improved synthesis: M. Blanc-Muesser, J. Defaye, Synthesis 1977, 568. Structure: S. F. Dyke, The Carbohydrates (Interscience, New York, 1960) p 45. Conformation: Reeves, J. Am. Chem. Soc. 72, 1499 (1950). Review: R. L. Whistler, M. L. Wolfrom, Methods in Carbohydrate Chemistry (Academic Press, New York, 1962) pp 140-145.