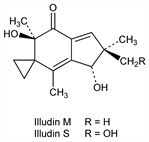
4899. Illudins
Description and references
Anti-tumor antibiotic substances produced by the poisonous basidiomycetes Clitocybe illudens (now called Omphalotus illudens): Anchel et al., Proc. Natl. Acad. Sci. USA 36, 300 (1950); 38, 927 (1952); and Lampteromyces japonicus: Nakanishi et al., Nature 197, 292 (1963); Endo et al., Chem. Commun. 1970, 309. Structure: McMorris, Anchel, J. Am. Chem. Soc. 87, 1594 (1965); Matsumoto et al., Tetrahedron 21, 2671 (1965); Tada et al., Chem. Pharm. Bull. 12, 853 (1964). Stereochemistry: Nakanishi et al., ibid. 856; Tetrahedron 21, 1231 (1965); Matsumoto et al., Bull. Chem. Soc. Jpn. 37, 1716 (1964). Abs config of illudin S: Harada, Nakanishi, Chem. Commun. 1970, 310. Total synthesis of illudin M: Matsumoto et al., J. Am. Chem. Soc. 90, 3280 (1968); Tetrahedron Lett. 1970, 1171; of illudin S: eidem, ibid. 1971, 2049. In vitro antitumor activity in human cancer cells: M. J. Kelner et al., J. Natl. Cancer Inst. 82, 1562 (1990).