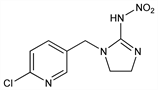
4910. Imidacloprid
Nomenclature
CAS number: 105827-78-9
1-[(6-Chloro-3-pyridinyl)methyl]-4,5-dihydro-N-nitro-1H-imidazol-2-amine; 1-[(6-chloro-3-pyridinyl)methyl]-N-nitro-2-imidazolidinimine; 1-(6-chloro-3-pyridylmethyl)-N-nitroimidazolidin-2-ylideneamine; 1-(2-chloro-5-pyridylmethyl)-2-(nitroimino)imidazolidine; BAY NTN 33893; Admire (Bayer CropSci.); Advantage (Bayer); Confidor (Bayer CropSci.); Gaucho (Gustafson); Marathon (Olympic); Merit (Bayer CropSci.); Premier (Bayer CropSci.); Provado (Bayer CropSci.); Trimax (Bayer CropSci.). C9H10ClN5O2; mol wt 255.66.
C 42.28%, H 3.94%, Cl 13.87%, N 27.39%, O 12.52%.
Description and references
Chloronicotinyl insecticide; targets the nicotinic acetylcholine receptor. Prepn: K. Shiokawa et al., EP 192060; eidem, US 4742060 (1986, 1988 both to Nihon Tokushu Noyaku Seizo KK). Properties and bioactivity: A. Elbert et al., Brighton Crop Prot. Conf. - Pests Dis. 1990, 21. Field trials: H. E. Schmeer et al., ibid. 29. LC-MS determn in fruits and vegetables: D. Zywitz et al., Dtsch. Lebensm. Rundsch. 99, 188 (2003). Review of mode of action and agricultural uses: W. Leicht, Pestic. Outlook 4, 17-21 (1993); of chemistry, toxicology, and veterinary use vs flea infestation: N. Mencke, P. Jeschke, Curr. Top. Med. Chem. 2, 701-715 (2002).