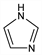4912. Imidazole
Nomenclature
CAS number: 288-32-4
1H-Imidazole; glyoxaline; 1,3-diazole; iminazole; miazole; pyrro[b]monazole; 1,3-diaza-2,4-cyclopentadiene. C3H4N2; mol wt 68.08.
C 52.93%, H 5.92%, N 41.15%.
Description and references
Prepd by the action of ammonia on glyoxal: Debus, Ann. 107, 204 (1858); from glyoxal, ammonia, and formaldehyde: Radziszewski, Ber. 15, 1493 (1882); Behrend, Schmitz, Ann. 277, 338 (1893); vapor phase synthesis from formamide and ethylenediamine in presence of a dehydrogenation catalyst: Green, US 3255200 (1966 to Air Products and Chemicals). Crystal structure: B. M. Craven et al., Acta Crystallogr. 33B, 2585 (1977). Acute toxicity: Nishie et al., Toxicol. Appl. Pharmacol. 14, 301 (1969). Review: Pyman, J. Soc. Dyers Colour. 36, 107 (1920). Monograph: K. Hofmann, Imidazole and Its Derivatives (Interscience, New York, 1953). Review of imidazole chemistry: Grimmett, Adv. Heterocycl. Chem. 12, 103-183 (1970).