4935. Indapamide
Nomenclature
CAS number: 26807-65-8
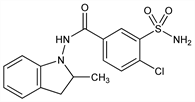
3-(Aminosulfonyl)-4-chloro-N-(2,3-dihydro-2-methyl-1H-indol-1-yl)benzamide; 4-chloro-N-(2-methyl-1-indolinyl)-3-sulfamoylbenzamide; N-(3-sulfamyl-4-chlorobenzamido)-2-methylindoline; S-1520; SE-1520; Bajaten (Merck KGaA); Fludex (Eutherapie); Indaflex (Lampugnani); Indamol (Aventis); Lozol (Aventis); Noranat (Novartis); Tandix (Biofarma); Veroxil (Baldacci). C16H16ClN3O3S; mol wt 365.83.
C 52.53%, H 4.41%, Cl 9.69%, N 11.49%, O 13.12%, S 8.77%.
Description and references
Prepn: Beregi et al., FR 2003311; eidem, US 3565911 (1969, 1971, both to Sci. Union et Cie, Soc. Franc. Recherche Med.). LC/MS determn in plasma: F. Albu et al., J. Chromatogr. B 816, 35 (2005). Pharmacology: Leary et al., Curr. Ther. Res. 15, 571 (1973); D. B. Campbell, R. A. Moore, Postgrad. Med. J. Suppl. 57, 7 (1981). Acute toxicity data: J. Kyncl et al., Arzneim.-Forsch. 25, 1491 (1975). Symposium on pharmacology and clinical efficacy: Am. J. Med. 84, Suppl. 1B, 1-111 (1988); Am. J. Cardiol. 65, Suppl. H, 1H-80H (1990). Comprehensive description: T. J. DiFeo, J. E. Shuster, Anal. Profiles Drug Subs. Excip. 23, 229-268 (1994).
Properties
Crystals from isopropanol/water, mp 160-162°. uv max (methanol): 242, 278, 286 nm (A1%1cm 630, 98, 100). Sol in methanol, acetic acid, ethyl acetate. Very slightly sol in chloroform. pKa (25°) 8.8 ± 0.2. LD50 in rats, mice, guinea pigs (mg/kg): 393-421, 410-564, 347-416 i.p.; 394-440, 577-635, 272-358 i.v.; >3000 all species orally (Kyncl).Derivative
Hemihydrate.Nomenclature
Damide (Benedetti); Ipamix (Gentili); Natrilix (Servier); Pressural (Polifarma).C16H16ClN3O3S.H2O; mol wt 374.84.
C 51.27%, H 4.57%, Cl 9.46%, N 11.21%, O 14.94%, S 8.55%.
Therapeutic Category
Antihypertensive; diuretic.
Keywords
Antihypertensive; Thiazides and Analogs; Diuretic; Thiazides and Analogs