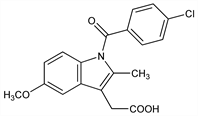
4968. Indomethacin
Nomenclature
Description and references
Nonsteroidal anti-inflammatory agent (NSAID); inhibits prostaglandin synthesis. Prepn: T. Y. Shen et al., J. Am. Chem. Soc. 85, 488 (1963); T. Y. Shen, US 3161654 (1964 to Merck & Co.). Alternate process: BE 679678 (1966 to Sumitomo). HPLC determn in plasma and urine: T. B. Vree et al., J. Chromatogr. 616, 271 (1993). Anti-inflammatory and antipyretic activity: C. A. Winter et al., J. Pharmacol. Exp. Ther. 141, 369 (1963). Metabolic studies: D. W. Yesair et al., Biochem. Pharmacol. 19, 1579 (1970). Toxicity: C. D. Klaassen, Toxicol. Appl. Pharmacol. 38, 127 (1976). Review of discovery, pharmacology and mechanism of action: T. Y. Shen, C. A. Winter in Advances in Drug Research vol. 12, A. B. Simmons, Ed. (Academic Press, New York, 1977) pp 89-245; of clinical trials in osteoarthritis: W. M. O'Brien, Clin. Pharmacol. Ther. 9, 94-107 (1968); of role in ductus closure: S. M. Douidar et al., Dev. Pharmacol. Ther. 11, 196-212 (1988).
Properties
Crystals exhibiting polymorphism, mp for one form ≈155°, for the other ≈162°. uv max (ethanol): 230, 260, 319 nm (ε 20800, 16200, 6290). pKa 4.5. Sol in ethanol, ether, acetone, castor oil. Practically insol in water. Stable in neutral or slightly acidic media; dec by strong alkali. LD50 i.p. in rats: 13 mg/kg (Klaassen).Derivative
Sodium salt trihydrate.Nomenclature
Properties
Pale yellow crystalline powder. pH of 1% soln: 8.4. Very sol in methanol; sol in water, ethanol. Very slightly sol in chloroform, acetone.Derivative
Meglumine salt.Nomenclature
Liometacen (Chiesi).Therapeutic Category
Anti-inflammatory; analgesic. For closure of patent ductus arteriosus in preterm infants.
Keywords
Anti-inflammatory (Nonsteroidal); Arylacetic Acid Derivatives; Analgesic; Non-opioids; Acetic/Propionic Acid Derivatives