4971. Indoxacarb
Nomenclature
CAS number: 173584-44-6
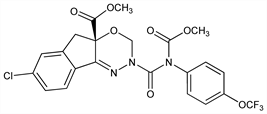
(4aS)-7-Chloro-2,5-dihydro-2-[[(methoxycarbonyl)[4-(trifluoromethoxy)phenyl]amino]carbonyl]indeno[1,2-e][1,3,4]oxadiazine-4a(3H)-carboxylic acid methyl
ester; DPX-KN128; Advion (DuPont); Avaunt (DuPont); Provaunt (DuPont); Steward (DuPont). C22H17ClF3N3O7; mol wt 527.83.
C 50.06%, H 3.25%, Cl 6.72%, F 10.80%, N 7.96%, O 21.22%.
Description and references
Oxadiazine pro-insectide; bioactivated in lepidoptera to the decarbomethoxylated metabolite that inhibits neuronal sodium channels. Prepn: G. D. Annis et al., WO 9211249 (1992 to DuPont); eidem, US 5462938 (1995). Physical properties and field trials: H. H. Harder et al., Brighton Crop Prot. Conf. - Pests Dis. 1996, 449. Efficacy of DPX-MP062, a 3:1 mixture of active (S)- to inactive (R)-isomer: U. Pluschkell et al., Pestic. Sci. 54, 85 (1998). Metabolism and mode of action: K. D. Wing et al., Crop Prot. 19, 537 (2000); B. Lapied et al., Br. J. Pharmacol. 132, 587 (2001).
Derivative
(RS)-Form.Nomenclature
CAS number: 144171-61-9
DPX-JW062.