4980. Insulin
Nomenclature
Description and references
Polypeptide hormone produced by pancreatic beta cells that regulates carbohydrate homeostasis. Converted by proteolysis from the single chain proinsulin, q.v., to the active dimer composed of 51 amino acid residues; mol wt ≈6000. Regulates carbohydrate and lipid metabolism, and influences protein synthesis. Insulin was the first protein for which the chemical structure and mol wt were determined. Also the first commercial health care product produced by recombinant DNA technology. Because of its solubility at physiological pH, insulin is rapidly absorbed after subcutaneous injection. Various complexes with protamine and/or zinc have been prepd to improve drug delivery. In addition to biological source (human, porcine or bovine), insulin formulations for therapeutic use are classified according to onset and duration of action. Isoln: F. G. Banting, C. H. Best, J. Lab. Clin. Med. 7, 251 (1921-22). Crystallization: Abel, Proc. Natl. Acad. Sci. USA 12, 132 (1926). Purification and properties: J. Lens, Biochim. Biophys. Acta 2, 76 (1948). Complete amino acid sequence of bovine insulin: F. Sanger, H. Tuppy, Biochem. J. 49, 463, 481 (1951); F. Sanger, E. O. P. Thompson, ibid. 53, 353, 366 (1953). Identification of 2 chain structure: A. P. Ryle et al., ibid. 60, 541 (1955). Review of structure determination: F. Sanger, Science 129, 1340 (1959). Structure of human insulin: D. Nichol, L. F. Smith, Nature 187, 483 (1960). Crystal structure: D. C. Hodgkin, Verh. Schweiz. Naturforsch. Ges. 150, 93 (1970). Synthesis of human insulin: P. G. Katsoyannis et al., J. Am. Chem. Soc. 88, 164, 166 (1966); by the enzymatic modification of porcine insulin: M. A. Ruttenberg, Science 177, 623 (1972). Review of synthetic insulins: P. G. Katsoyannis, Recent Prog. Horm. Res. 23, 505-563 (1967). Synthesis of human insulin gene: H. M. Hsiung et al., Nucleic Acids Res. 6, 1371 (1979); 7, 2199 (1979); 8, 5753 (1980); S. A. Narang et al., Nucleic Acids Symp. Ser. 7, 377 (1980). Review of the development and production of human insulin by recombinant DNA technology: I. S. Johnson, Science 219, 632-637 (1983). Molecular basis of insulin action: M. P. Czech, Annu. Rev. Biochem. 46, 359 (1977). History: M. Bliss, The Discovery of Insulin (Univ. Chicago Press, Chicago, 1982) 304 pp. Review of biosynthesis: D. F. Steiner et al., Recent Prog. Horm. Res. 25, 207-282 (1969). Review of the structure and function of the insulin receptor: J. Lee, P. F. Pilch, Am. J. Physiol. 266, C319-C334 (1994). Symposium on the physiological regulation of insulin secretion and the pathogenesis of diabetes: Diabetologia 37, Suppl. 2, S1-S187 (1994). Review of bioactivity, pharmacokinetics and therapeutic efficacy of human insulin: R. N. Brogden, R. C. Heel, Drugs 34, 350-371 (1987). Review of insulin formulations and therapy: J. A. Galloway, R. E. Chance, Horm. Metab. Res. 26, 591-598 (1994); of analogs and alternative delivery methods: J. E. Gerich, Am. J. Med. 113, 308-316 (2002); of clinical development of inhaled formulations: L. D. Mastrandrea, T. Quattrin, Adv. Drug Delivery Rev. 58, 1061-1075 (2006).
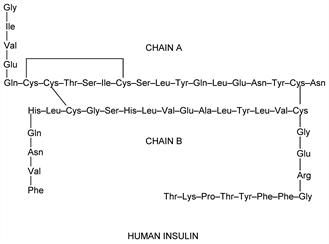
Properties
Crystals, hexagonal system, usually obtained as flat rhombohedra and contg 0.4% Zn. Readily sol in dil acids and alkalies. Isoelectric point 5.30 to 5.35.Derivative
Bovine insulin.Nomenclature
Derivative
Porcine insulin.Nomenclature
Description and references
Differs from human insulin by a single amino acid substitution.
Derivative
Recombinant human insulin.Nomenclature
Biosynthetic human insulin; insulin (prb); Huminsulin (Lilly); Humulin (Lilly); Insuman Rapid (Sanofi-Aventis); Velosulin (Novo).Description and references
Human insulin prepd by recombinant DNA technology. Clinical evaluation of inhaled intrapulmonary delivery in type 1 diabetes: J. S. Skyler et al., Lancet 357, 331 (2001); in type 2 diabetes: W. T. Cefalu et al., Ann. Intern. Med. 134, 203 (2001).
Derivative
Semi-synthetic human insulin.Nomenclature
Insulin (emp); Actrapid (Novo); Exubera (Pfizer); Humalog (Lilly); Novolin (Novo).Properties
Human insulin prepd by enzymatic modification of porcine insulin.Derivative
Zinc insulin.Nomenclature
Description and references
Crystalline prepn of insulin containing 0.45-0.9% zinc. Formulated as suspensions in physiological saline; size of the particles determines the duration of action. Formulations are designated as prompt (or semilente), lente and extended (or ultralente).
Derivative
Protamine zinc insulin.Nomenclature
Description and references
Suspensions of insulin modified by the addition of zinc chloride and protamine sulfate.
Properties
White or almost white suspension, pH 7.1-7.4. Onset of action occurs from 4-6 hrs after s.c. injection; duration of action is 36 hrs.Derivative
Isophane insulin.Nomenclature
NPH insulin; neutral protein Hagedorn insulin; Insulatard (Novo).Description and references
Crystallized prepn of protamine, zinc and insulin. Prepn: H. C. Hagedorn et al., J. Am. Med. Assoc. 106, 177 (1936). Review: P. Felig, ibid. 251, 393-396 (1984).
Properties
White suspensions of rod-shaped crystals ≈30 nm in length, pH 7.1-7.4. Onset of action is 3-4 hrs following s.c. injection; duration of action is 18-28 hrs.Derivative
Insulin 131I.Nomenclature
Radio-iodinated insulin.Description and references
Prepn: Burrows et al., J. Clin. Invest. 36, 393 (1957); Grodsky et al., Arch. Biochem. Biophys. 81, 264 (1959).
Use
Insulin 131I used in the study of insulin binding factors from insulin resistant sera.Therapeutic Category
Antidiabetic.
Therapeutic Category (Veterinary)
Antidiabetic.Keywords
Antidiabetic; Hormones/Analogs