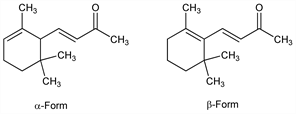
5056. Ionone
Nomenclature
Irisone (Givaudan).Description and references
Mixture in which the major constituents are α-ionone, (E)-4-(2,6,6-trimethyl-2-cyclohexen-1-yl)-3-buten-2-one, and β-ionone, (E)-4-(2,6,6-trimethyl-1-cyclohexen-1-yl)-3-buten-2-one. The latter is a key intermediate in the synthesis of vitamin A. Isoln from the volatile oil of Boronia megastigma Nees., Rutaceae: Naves, Parry, Helv. Chim. Acta 30, 419 (1946). Prepn by condensing citral with acetone: Tremann, Schmidt, Ber. 33, 3703 (1900); Hibbert, Cannon, J. Am. Chem. Soc. 46, 119 (1924); Krishna, Joshi, J. Org. Chem. 22, 224 (1957). Prepn from isobutylene and vinyl methyl ketone: Pasedach, Seefelder, DE 1000374 (1957 to BASF), C.A. 54, 1595g (1960). Prepn by cyclization of pseudoionone with H2SO4: Kimel et al., J. Org. Chem. 22, 1611 (1957); Kaiser, Kimel, US 2877271 (1959 to Hoffmann-La Roche). Review: Naves, J. Soc. Cosmet. Chem. 22, 439 (1971).