5078. Iproniazid
Nomenclature
CAS number: 54-92-2
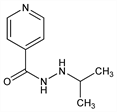
4-Pyridinecarboxylic acid 2-(1-methylethyl)hydrazide; isonicotinic acid 2-isopropylhydrazide; 1-isonicotinoyl-2-isopropylhydrazine; 1-isonicotinyl-2-isopropylhydrazine. C9H13N3O; mol wt 179.22.
C 60.31%, H 7.31%, N 23.45%, O 8.93%.
Description and references
Monoamine oxidase inhibitor. Prepd by treating isoniazid with isopropyl bromide in abs alcohol in the presence of sodium: McMillan et al., J. Am. Pharm. Assoc. Sci. Ed. 42, 457 (1953). Alternate synthesis: Fox, Gibas, J. Org. Chem. 18, 994 (1953). Comprehensive description: F. Belal, H. Abdel-Aliem, Anal. Profiles Drug Subs. 20, 337-368 (1991).
Properties
Needles from benzene + ligroin, mp 112.5-113.5°. Freely sol in water, alcohol; pH of aq solns about 6.7.Derivative
Dihydrochloride.C9H13N3O.2HCl; mol wt 252.14.
C 42.87%, H 6.00%, N 16.67%, O 6.35%, Cl 28.12%.
Properties
Rhomboid crystals from isopropanol, dec at 227-228°. Freely sol in water; aq solns are very acidic.Derivative
Phosphate.Nomenclature
CAS number: 305-33-9
Marsilid (Roche). C9H13N3O.H3PO4; mol wt 277.21.
C 38.99%, H 5.82%, N 15.16%, O 28.86%, P 11.17%.
Properties
White to slightly yellowish powder, melting range 175-184°. pH of a 5% soln: 2.7-3.6. uv spectrum: J. Kracmar, J. Kracmarova, Pharmazie 34, 27 (1979). uv max (methanol): 265 nm (A1%1cm 166); (water): 264 nm (A1%1cm 176); (0.1N HCl): 267 nm (A1%1cm 179); (0.1N KOH): 244, 272, 308 nm (A1%1cm 113, 121, 135). Soly at 25° (mg/ml): water ≈188; methanol ≈21; ethanol (96%) ≈9; chloroform ≈0.6; diethyl ether <0.1; hexane <0.1.Therapeutic Category
Antidepressant.
Keywords
Antidepressant; Hydrazides/Hydrazines; Monoamine Oxidase Inhibitor