5090. Irigenin
Nomenclature
CAS number: 548-76-5
5,7-Dihydroxy-3-(3-hydroxy-4,5-dimethoxyphenyl)-6-methoxy-4H-1-benzopyran-4-one; 3′,5,7-trihydroxy-4′,5′,6-trimethoxyisoflavone. C18H16O8; mol wt 360.31.
C 60.00%, H 4.48%, O 35.52%.
Description and references
From rhizome of Iris florentina L., Iridaceae: de Laire, Tiemann, Ber. 26, 2010 (1893). Structure: Baker, J. Chem. Soc. 1928, 1022. Synthesis: Baker et al., Tetrahedron Lett. no. 5, 6 (1960); Farkas, Várady, Ber. 93, 2685 (1960); Baker et al., J. Chem. Soc. C 1970, 1219.
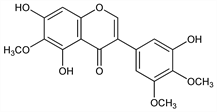
Properties
Yellow plates or needles from dil alc, mp 185°. uv max (abs ethanol): 267 nm. Sol in warm alcohol, benzene, chloroform; practically insol in water, ether, petr ether.Derivative
Triacetate.C24H22O11; mol wt 486.42.
C 59.26%, H 4.56%, O 36.18%.
Properties
Prisms from dil acetic acid, mp 127-128°.Derivative
7-Glucoside.Nomenclature
Iridin; 7-(glucosyloxy)-3′,5-dihydroxy-4′,5′,6-trimethoxyisoflavone.C24H26O13; mol wt 522.46.
C 55.17%, H 5.02%, O 39.81%.