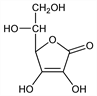
5126. Isoascorbic Acid
Nomenclature
CAS number: 89-65-6
d-erythro-Hex-2-enonic acid γ-lactone; d-araboascorbic acid; erythorbic acid; isovitamin C; saccharosonic acid; glucosaccharonic acid; d-erythro-3-ketohexonic acid lactone; d-erythro-3-oxohexonic acid lactone; erycorbin; Mercate “5”; Neo-Cebicure. C6H8O6; mol wt 176.12.
C 40.92%, H 4.58%, O 54.51%.
Description and references
Epimer of l-ascorbic acid. Has one-twentieth of the vitamin C activity of l-ascorbic acid. Prepd by treating methyl 2-keto-d-gluconate with sodium methoxide: Maurer, Schiedt, Ber. 66, 1054 (1933); 67, 1239 (1934); Ohle et al., ibid. 67, 324 (1934); Baird et al., J. Chem. Soc. 1934, 63; Reichstein et al., Helv. Chim. Acta 17, 516 (1934). Synthesis from sucrose: Heimann, Reiff, Pharm. Zentralhalle 93, 97 (1954). Production by Penicillium spp: Takahashi et al., Nature 188, 411 (1960); US 3052609 (1962 to Sankyo); Bull. Agric. Chem. Soc. Jpn. 24, 533 (1960), C.A. 55, 1788 (1961). Conformation: Matsui et al., Agric. Biol. Chem. 27, 185 (1963).