5217. Isoproterenol
Nomenclature
Description and references
β-Adrenergic agonist. Catecholamine derivative prepd by the reaction of 3,4-dihydroxy-α-haloacetophenone with an excess of isopropylamine: Scheuing, Thom, DE 723278 (1942 to Boehringer, Ing.); US 2308232 (1943); from guaiacol and chloral hydrate: Beke et al., Pharm. Zentralhalle 92, 237 (1953). Resolution: Kerschbaum, Benedikt, Monatsh. Chem. 83, 1090 (1952); Beccari et al., Science 118, 249 (1953); Delmar et al., US 2715141 (1955 to Delmar Chem.). Configuration: Pratesi et al., Farmaco Ed. Sci. 15, 3 (1960). Toxicity: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Effect on gastric acid secretion: M. J. Daly, Scand. J. Gastroenterol. 89, 3 (1984). Use in treatment of primary pulmonary hypertension: D. A. Pietro et al., N. Engl. J. Med. 310, 1032 (1984). Review of pharmacology and comparison with other β-adrenoceptor agonists: V. T. Popa, J. Asthma 21, 183-207 (1984). Comprehensive description: M. Tariq, A. A. Al-Badr, Anal. Profiles Drug Subs. 14, 391-422 (1985).
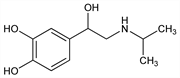
Derivative
dl-Form.Properties
Crystals from alc, mp 155.5°. pKa 8.64. LD50 in male, female rats (mg/kg): 3675, 4282 orally (Goldenthal).Derivative
dl-Form hydrochloride.Nomenclature
Aerolone (Lilly); Aerotrol (Abbott); Euspiran; Isomenyl (Kaken); Isovon; Mistarel; Suscardia (Pharmax).Properties
Crystals from alc, mp 170-171°. Soly: one gram dissolves in 3 ml water, in 50 ml ethanol (95%). Less sol in abs ethanol. Practically insol in chloroform, ether, benzene. pH of 1% aq soln about 5. Aq solns turn brownish-pink upon prolonged exposure to air or upon addition of alkali. LD50 orally in rats: 2221 ±93 mg/kg (Goldenthal).Derivative
dl-Form sulfate dihydrate.Nomenclature
Properties
Crystals from acetone + methanol, mp 128° (some decomp). One gram dissolves in about 4 ml water. Slightly sol in alc. Practically insol in chloroform, ether, benzene. pH of a 1% aq soln about 5.Derivative
l-Form.Properties
Crystals, mp 164-165°. [α]D19 45.0° (c = 2 in 2N HCl).Derivative
l-Form hydrochloride.Properties
Crystals, dec 162-164°. [α]D20 50°.Derivative
l-Form d-bitartrate dihydrate.Nomenclature
Isolevin.Properties
Crystals, mp 80-83° (sinters at 78°). [α]D19 14.9° (c = 2.31).Therapeutic Category
Bronchodilator.
Therapeutic Category (Veterinary)
Sympathomimetic. Chiefly as bronchodilator.Keywords
β-Adrenergic Agonist; Bronchodilator; Ephedrine Derivatives