5252. Jacobsen's Catalyst
Nomenclature
CAS number: 149656-63-3
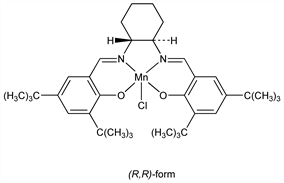
(SP-5-13)-Chloro[[rel-2,2′-[(1R,2R)-1,2-cyclohexanediylbis[(nitrilo-κN)methylidyne]]bis[4,6-bis(1,1′-dimethylethyl)phenolato-κO]](2)]manganese. C36H52ClMnN2O2; mol wt 635.20.
C 68.07%, H 8.25%, Cl 5.58%, Mn 8.65%, N 4.41%, O 5.04%.
Description and references
Manganese-salen Schiff base complex used as a catalyst in asymmetric syntheses. Prepn: E. N. Jacobsen et al., J. Am. Chem. Soc. 113, 7063 (1991); large-scale: J. F. Larrow et al., J. Org. Chem. 59, 1939 (1994). Synthesis and enantioselectivity of dimer: K. B. M. Janssen et al., Tetrahedron: Asymmetry 8, 3481 (1997). HPLC determn of enantiomeric purity in commercial samples: J. Zukowski, Chirality 10, 362 (1998). Mechanism of asymmetric epoxidation of indenes: D. L. Hughes et al., J. Org. Chem. 62, 2222 (1997). Use in epoxidation of isoflavones: W. Adam et al., Tetrahedron: Asymmetry 9, 1121 (1998).
Derivative
(R,R)-form.Nomenclature
CAS number: 138124-32-0
(R,R)-[N,N′-Bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediamine]manganese (III) chloride.