5299. Kethoxal
Nomenclature
CAS number: 27762-78-3
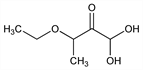
3-Ethoxy-1,1-dihydroxy-2-butanone; β-ethoxy-α-ketobutyraldehyde monohydrate; 3-ethoxy-2-oxobutyraldehyde hydrate; U-2032. C6H12O4; mol wt 148.16.
C 48.64%, H 8.16%, O 43.19%.
Description and references
Antiviral glyoxal derivative. Specifically reacts with unpaired guanine residues of nucleic acids. Prepn: L. Rappen, J. Prakt. Chem. 157, 177 (1941); and antiviral activity: B. D. Tiffany et al., J. Am. Chem. Soc. 79, 1682 (1957). Reaction with guanine residues: M. Staehelin, Biochim. Biophys. Acta 31, 448 (1959); R. Shapiro, J. Hachmann, Biochemistry 5, 2799 (1966). Use as RNA probe: M. Litt, V. Hancock, ibid. 6, 1848 (1967); M. Balzer, R. Wagner, Anal. Biochem. 256, 240 (1998). Use in peptide mapping to identifiy arginine residues: O. T. Akinsiku et al., J. Mass Spectrom. 40, 1372 (2005).
Properties
Yellow oil. bp11 54-58°. bp760 145°. Initial nD23.7 1.4348. Miscible with alc; sol in benzene with yellow color; one part dissolves in ten parts of water.Derivative
Bis(thiosemicarbazone).Nomenclature
CAS number: 2507-91-7
2,2′-[1-(1-Ethoxyethyl)-1,2-ethanediylidene]bishydrazinecarbothioamide; gloxazone; BW-356-C-61; NSC-82116. C8H16N6OS2; mol wt 276.38.
C 34.77%, H 5.84%, N 30.41%, O 5.79%, S 23.20%.