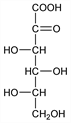
5304. 2-Keto-l-gulonic Acid
Nomenclature
Description and references
Important intermediate in vitamin C manufacture. Prepn from sorbitol: Reichstein, Grüssner, Helv. Chim. Acta 17, 311 (1934); Reichstein, US 2301811 (1942); from l-sorbose: Haworth et al., GB 443901 (1936); by microbial conversion: Huang, US 3043749 (1962 to Pfizer); Motizuki et al., US 3234105 (1966 to Takeda); Z. Y. Yuan et al., Ann. N.Y. Acad. Sci. 672, 628 (1992). Biosynthesis and metabolism in bacteria: S. Makover et al., Biotechnol. Bioeng. 17, 1485 (1975). NMR structure determn: T. C. Crawford et al., J. Am. Chem. Soc. 102, 2220 (1980). HPLC separation in fermentation broth: R. A. Lazarus, J. L. Seymour, Anal. Biochem. 157, 360 (1986). Review of early syntheses: T. C. Crawford, S. A. Crawford, Adv. Carbohydr. Chem. Biochem. 37, 79-159 (1980).