5306. Ketorolac
Nomenclature
Description and references
Prostaglandin biosynthesis inhibitor. Prepn and separation of isomers: BE 856681; J. M. Muchowski, A. F. Kluge, US 4089969 (both 1978 to Syntex). Alternate processes: J. M. Muchowski, R. Greenhouse, US 4347186 (1982 to Syntex); F. Franco et al., J. Org. Chem. 47, 1682 (1982); J. B. Doherty, US 4496741 (1985 to Merck & Co.). Absolute configuration: A. Guzman et al., J. Med. Chem. 29, 589 (1986). Structure-activity relationships: J. M. Muchowski et al., ibid. 28, 1037 (1985). Pharmacology and analgesic, anti-inflammatory profile of ketorolac and its tromethamine salt: W. H. Rooks et al., Agents Actions 12, 684 (1982); eidem, Drugs Exp. Clin. Res. 11, 479 (1985). Clinical comparison with acetaminophen in post-operative pain: H. J. McQuay et al., Clin. Pharmacol. Ther. 39, 89 (1986).
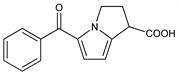
Properties
Crystals from ethyl acetate + ether, mp 160-161°. uv max in methanol: 245, 312 nm (ε 7080, 17400). pKa 3.49 ±0.02. LD50 orally in mice: ≈200 mg/kg (Rooks).Derivative
(±)-Form tromethamine salt.Nomenclature
Derivative
(+)-Form.Properties
Crystals from hexane + ethyl acetate, mp 174° (Guzman); also reported as mp 154-156° (Muchowski, Kluge). [α]D +173° (c = 1 in methanol).Derivative
()-Form.Properties
Crystals from hexane + ethyl acetate, mp 169-170° (Guzman); also reported as mp 153-155° (Muchowski, Kluge). [α]D 176° (c = 1 in methanol).Therapeutic Category
Analgesic; anti-inflammatory.
Keywords
Analgesic; Non-opioids; Anti-inflammatory (Nonsteroidal); Arylcarboxylic Acids