5309. Khellin
Nomenclature
Description and references
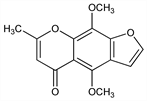
Found in seeds of Ammi visnaga Lam., Umbelliferae; one of the active principles of the Egyptian traditional medicine known as “khella.” See also: visnadine, visnagin. Isoln and structure: Spth, Gruber, Ber. 71, 106 (1938). Synthesis: Baxter et al., J. Chem. Soc. 1949, S 30; Gardner et al., J. Org. Chem. 15, 841 (1950); Schnberg, Sina, J. Am. Chem. Soc. 72, 1611, 3396 (1950); M. W. Reed, H. W. Moore, J. Org. Chem. 53, 4166 (1988). Crystal structure: J. P. Beale, Cryst. Struct. Commun. 2, 125 (1973). Toxicity data: E. Busch et al., Arzneim.-Forsch. 11, 915 (1961). GC determn in serum: A. S. Carlin et al., J. Chromatogr. 614, 324 (1993). Clinical evaluation in vitiligo: B. Ortel et al., J. Am. Acad. Dermatol. 18, 693 (1988). Comprehensive description: M. A. Hassan, M. U. Zubai, Anal. Profiles Drug Subs. 9, 371-396 (1980).
Properties
Crystals from methanol. Bitter taste. mp 154-155°. bp0.05 180-200°. uv max (alc): 250, 338 nm (E1%1cm 1600, 200). Soly in g/100 ml at 25°: water 0.025; acetone 3.0; methanol 2.6; isopropanol 1.25; ether 0.5. Much more sol in hot water and hot methanol. LD50 in mice, rats (mg/kg): 30.6, 34.4 i.v.; 50.8, 68.8 orally (Busch).Therapeutic Category
Vasodilator (coronary). Photosensitizer in treatment of vitiligo.
Keywords
Vasodilator (Coronary)