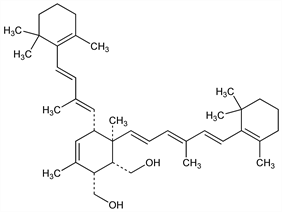
5315. Kitol
Nomenclature
CAS number: 4626-00-0
[1α,2α,5α(1E,3E),6β(1E,3E,5E)]-3,6-Dimethyl-5-[2-methyl-4-(2,6,6-trimethyl-1-cyclohexen-1-yl)-1,3-butadienyl]-6-[4-methyl-6-(2,6,6-trimethyl-1-cyclohexen-1-yl)-1,3,5-hexatrienyl]-3-cyclohexene-1,2-dimethanol. C40H60O2; mol wt 572.90.
C 83.86%, H 10.56%, O 5.59%.
Description and references
One of the provitamins A. Obtained from mammalian liver oil: Embree, Shantz, J. Am. Chem. Soc. 65, 910 (1943); Clough et al., Science 105, 436 (1947); Barua, Morton, Biochem. J. 45, 309 (1949); Chatan, Fridenson, Compt. Rend. 234, 1094 (1952). Purification: Tawara, Fukazawa, C.A. 47, 12483e (1953). Conversion to vitamin A: Libermann, Grundland, Compt. Rend. 224, 1033 (1947). Structure: Burger et al., Chem. Commun. 1965, 588; Giannotti et al., ibid. 1966, 28; Giannotti et al., Bull. Soc. Chim. Fr. 1966, 3299; Burger, Garbers, J. Chem. Soc. Perkin Trans. 1 1973, 590.