5345. Lactucin
Nomenclature
CAS number: 1891-29-8
[3aR-(3aα,4β,9aα,9bβ)]-3,3a,4,5,9a,9b-Hexahydro-4-hydroxy-9-(hydroxymethyl)-6-methyl-3-methyleneazuleno[4,5-b]furan-2,7-dione. C15H16O5; mol wt 276.28.
C 65.21%, H 5.84%, O 28.96%.
Description and references
From various Lactuca spp and Cichorium intybus L., Compositae. Isoln: Schenck, Graf, Arch. Pharm. 274, 537 (1936); 275, 36 (1937); Schenck et al., ibid. 294, 17 (1961). Purification: Spth et al., Monatsh. Chem. 82, 114 (1951). Structure: Dolejs et al., Collect. Czech. Chem. Commun. 23, 2195 (1958); Barton, Narayanan, J. Chem. Soc. 1958, 963; Michl, Hgenauer, Monatsh. Chem. 89, 317 (1958). Revised stereochemistry: Bachelor, It, Can. J. Chem. 51, 3626 (1973).
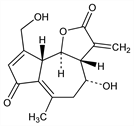
Properties
Crystals from methanol, sinters at 218°, mp 228-233°. [α]D +49° (c = 0.90 in methanol), +77.9° (c = 3.44 in pyridine). uv max: 257 nm (ε 14,000). Soluble in water, ethanol, methanol, ethyl acetate, dioxane anisol.Derivative
p-Hydroxyphenylacetate hydrate.Nomenclature
Intybin; lactucopicrin.C23H22O7; mol wt 410.42.
C 67.31%, H 5.40%, O 27.29%.
Description and references
Identity with intybin: Schmitt, Bot. Arch. 40, 516 (1940), C.A. 36, 5616 (1942). Structure: Michl, Hgenauer, Monatsh. Chem. 91, 500 (1960).