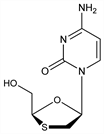
5352. Lamivudine
Nomenclature
Description and references
Reverse transcriptase inhibitor. Prepn: J. A. V. Coates et al., WO 9117159 C.A. 117, 111989 (1991). Synthesis of enantiomers: J. W. Beach et al., J. Org. Chem. 57, 2217 (1992); of ()-enantiomer: D. C. Humber et al., Tetrahedron Lett. 33, 4625 (1992). HPLC determn in urine: D. M. Morris, K. Selinger, J. Pharm. Biomed. Anal. 12, 255 (1994). Clinical trial in hepatitis B: F. Nevens et al., Gastroenterology 113, 1258 (1997). Review of pharmacology and clinical efficacy in HIV infection: C. M. Perry, D. Faulds, Drugs 53, 657-680 (1997); of clinical experience in chronic hepatitis B and cirrhosis: C. Hanché, J-P Villeneuve, Expert Opin. Pharmacother. 7, 1835-1843 (2006).
Properties
Crystals from boiling ethanol, mp 160-162°. [α]D21 135° (c = 0.38 in methanol). Soly in water (20°): ≈70 mg/ml.Therapeutic Category
Antiretroviral.
Keywords
Reverse Transcriptase Inhibitor; Antiretroviral; Reverse Transcriptase Inhibitors; Nucleosides/Nucleotides