5354. Lanatosides
Description and references
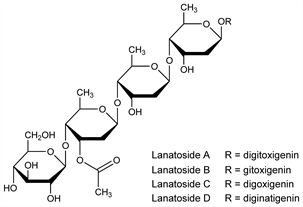
Family of four glycosides, A, B, C, D, isolated from various species of Digitalis including D. lanata Ehrh., Scrophulariaceae: Stoll, Kreis, Helv. Chim. Acta 16, 1049 (1933); Ligeti, Pharmazie 12, 433 (1957); 14, 162 (1959); Angliker et al., Ann. 607, 131 (1957); D. Lutea L., Scrophulariaceae: Cole, Gisvold, J. Am. Pharm. Assoc. Sci. Ed. 47, 654 (1958). Structure: Tschesche et al., Ber. 92, 2258 (1959); Uskert, Ann. 638, 199 (1960); Kuhn et al., Helv. Chim. Acta 45, 881 (1962). Absorption spectrum: Bell, J. Am. Pharm. Assoc. Sci. Ed. 49, 277 (1960). Cardioactivity of lanatoside C: K.-O. Haustein, Pharmacology 11, 117 (1974). Mitogenicity: L. L. G. Hammarstrm, C. I. E. Smith, J. Immunol. 120, 694 (1978). HPLC determn: V. Y. Davydov et al., J. Chromatogr. 248, 49 (1982); of lanatoside C: F. Orosz et al., Anal. Biochem. 156, 171 (1986).
Derivative
Lanatoside A.Nomenclature
Properties
Long, flat prisms from methanol, dec 245-248°. [α]D20 +31.6° (0.48 g in 25 ml 95% alc); [α]D20 +23.2° (0.95 g in 25 ml dioxane). Soluble in 20 parts methanol, 40 parts alcohol, 225 parts chloroform, 16,000 parts water.Derivative
Lanatoside B.Nomenclature
Properties
Long, flat prisms from alcohol, dec 245-248° after drying in high vacuum at 150°. [α]D20 +36.7° (0.47 g in 25 ml 95% alc); [α]D20 +31.8° (0.25 g in 14.1 ml dioxane). Soluble in 20 parts methanol, 40 parts alcohol, 550 parts chloroform. Nearly insol in water.Derivative
Lanatoside C.Nomenclature
Properties
Long, flat prisms from alcohol, dec 248-250°, after drying in vacuum at 150°. [α]D20 +33.4 to +33.7° (200 mg dry weight in 10 ml alcohol). One gram dissolves in 20,000 ml methanol, in 2000 ml chloroform. Freely sol in pyridine, dioxane. Practically insol in ether, petr ether.Derivative
Lanatoside D.Nomenclature
Properties
Needles from methanol and water, dec 242-250°. [α]D20 +40.5° (c = 5.95 in methanol). uv max: 220 nm (log ε 4.16).Therapeutic Category
Cardiotonic.
Keywords
Cardiotonic