5378. Latrunculins
Description and references
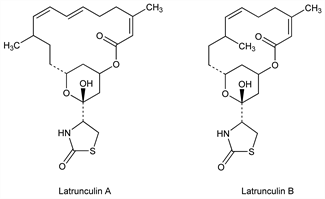
Highly potent toxins which disrupt microfilament organization by binding the actin monomer. Isolated from the Red Sea sponge Latrunculia magnifica Keller; also found in the Pacific nudibranch Chromodoris elisabethina and the Fijian sponge Spongia mycofijiensis. Isoln and biological activities: I. Néeman et al., Mar. Biol. 30, 293 (1975). First family of natural products possessing the 2-thiazolidinone moiety; latrunculins A-D are 14- or 16- membered macrolides, infrastructures previously unknown in marine isolates. Structure elucidation of A and B: A. Groweiss et al., J. Org. Chem. 48, 3512 (1983); of A through D: Y. Kashman et al., Tetrahedron 41, 1905 (1985). Chemistry of A and B: D. Blasberger et al., Ann. 1989, 1171. Total synthesis of B: R. Zibuck et al., J. Am. Chem. Soc. 108, 2451 (1986); of A: J. D. White, M. Kawasaki, J. Am. Chem. Soc. 112, 4991 (1990); of A, B, C and M: A. B. Smith, III et al., ibid. 114, 2995 (1992). Comparative study with cytochalasin D, q.v., on the effects actin organization and cell processes: I. Spector et al., Cell Motil. Cytoskeleton 13, 127 (1989). Inhibition of the actin monomer: W. M. Morton et al., Nat. Cell Biol. 2, 376 (2000). Review of use as research tool: K. Ayscough, Methods Enzymol. 298, 18-25 (1998).
Derivative
Latrunculin A.Nomenclature
Properties
Foam. [α]D24 +152° (c = 1.2 in chloroform). uv max (methanol): 218 nm (ε 23500).Derivative
Latrunculin B.Nomenclature
Description and references
Major toxin. Localization within the Red Sea sponge: O. Gillor et al., Mar. Biotechnol. 2., 213 (2000).