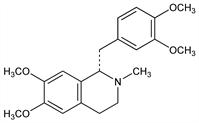
5381. Laudanosine
Nomenclature
CAS number: 2688-77-9
(1S)-1-[(3,4-Dimethoxyphenyl)methyl]-1,2,3,4-tetrahydro-6,7-dimethoxy-2-methylisoquinoline; 1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-veratrylisoquinoline; N-methyltetrahydropapaverine. C21H27NO4; mol wt 357.44.
C 70.56%, H 7.61%, N 3.92%, O 17.90%.
Description and references
Occurs in opium (0.0008%). It is the last alkaloid to be separated from morphine extraction mother liquors; occurs as (+)-form. Synthesis: Pictet, Finkelstein, Ber. 42, 1979 (1909); Frydman et al., Tetrahedron 4, 342 (1958); Elliott, J. Heterocycl. Chem. 9, 853 (1972). Asymmetric synthesis: M. Konda et al., Chem. Pharm. Bull. 23, 1025 (1975); of (R)-()-laudanosine: M. Konda et al., ibid. 25, 69 (1977); R. E. Gawley, G. A. Smith, Tetrahedron Lett. 29, 301 (1988). Configuration: Leithe, Ber. 64, 2827 (1931); Faltis, Adler, Arch. Pharm. 284, 281 (1951); Corrodi, Hardegger, Helv. Chim. Acta 39, 889 (1956).