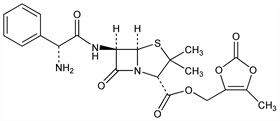
5437. Lenampicillin
Nomenclature
CAS number: 86273-18-9
(2S,5R,6R)-6-[[(2R)-Aminophenylacetyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid (5-methyl-2-oxo-1,3-dioxol-4-yl)methyl ester; ampicillin (5-methyl-2-oxo-1,3-dioxolen-4-yl)methyl ester; 6-[d()-α-aminophenylacetamido]penicillanic acid (5-methyl-2-oxo-1,3-dioxol-4-yl)methyl
ester. C21H23N3O7S; mol wt 461.49.
C 54.65%, H 5.02%, N 9.11%, O 24.27%, S 6.95%.
Description and references
Orally active ampicillin prodrug. Prepn: F. Sakamoto et al., EP 39086; eidem, US 4342693 (1981, 1982 both to Kanebo); F. Sakamoto et al., Chem. Pharm. Bull. 32, 2241 (1984); S. Ikeda et al., ibid. 4316. Metabolism in man, dogs, rats: N. Awata et al., Jpn. J. Antibiot. 38, 1776, 1785, C.A. 104, 161479u, 122550r (1985). Human pharmacokinetics: A. Saito, M. Nakashima, Antimicrob. Agents Chemother. 29, 948 (1986). Series of articles on antibacterial activity, mutagenicity, pharmacology, clinical trials: Chemotherapy (Tokyo) 32, Suppl. 8, pp 1-772 (1984). Acute toxicity data: F. Ogino et al., ibid. 31.
Derivative
Hydrochloride.Nomenclature
CAS number: 80734-02-7
KB-1585; KBT-1585; Takacillin (Torii); Varacillin (Kanebo). C21H23N3O7S.HCl; mol wt 497.95.
C 50.65%, H 4.86%, N 8.44%, O 22.49%, S 6.44%, Cl 7.12%.
Properties
Crystals from isopropanol-ethyl acetate, mp 145° (dec). LD50 in male, female rats, male, female mice (mg/kg): approx 10000, approx 10000, 8294, 8492 orally; 4362, 4471, 3576, 4284 s.c.; 876, 838, 711, 775 i.v.; in dogs (mg/kg): >300 orally (Ogino).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins