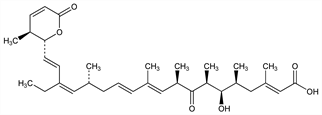
5444. Leptomycin B
Nomenclature
Description and references
Antibiotic compound with antifungal and antitumor activity. Inhibits the CRM1/exportin1 pathway of nuclear transport in eukaryotic cells, facilitating its use in the study of nuclear export. Isoln from Streptomyces sp. ATS1287: T. Hamamoto et al., J. Antibiot. 36, 639 (1983); from actinomycete ATCC 39366: G. C. Hokanson et al., EP 139458; eidem, US 4771070 (1985, 1988 both to Warner Lambert). Antitumor activity: B. J. Roberts et al., Cancer Chemother. Pharmacol. 16, 95 (1986); and antimicrobial activity: J. B. Tunac et al., J. Antibiot. 38, 460 (1985). Synthesis and abs config: M. Kobayashi et al., Tetrahedron Lett. 39, 8291 (1998). HPLC determn in fermentation extracts: M. Stadler et al., J. Chromatogr. A 818, 187 (1998). Review of use as inhibitor of nuclear export: M. Yoshida et al., Actinomycetologica 12, 120-128 (1998).