5464. Levodopa
Nomenclature
Description and references
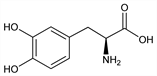
Naturally occurring form of dopa, q.v., the biological precursor of the catecholamines. Prepn from l-3-nitrotyrosin: Wasser, Lewandowski, Helv. Chim. Acta 4, 657 (1921); from 3-(3,4-methylenedioxyphenyl)-l-alanine: Yamada et al., Chem. Pharm. Bull. 10, 693 (1962); from l-tyrosine: Vorbrüggen, Krolikiewicz, Ber. 105, 1168 (1972); Bretschneider et al., Helv. Chim. Acta 56, 2857 (1973); from Vicia faba beans: Wysong, US 3253023 (1966 to Dow Chem.); by fermentation of l-tyrosine: Sih et al., J. Am. Chem. Soc. 91, 6204 (1969); Florent, Renaut, DE 2102793 (1971 to Rhne-Poulenc), C.A. 75, 108505f (1971). Sepn from racemate: Vogler, Baumgartner, Helv. Chim. Acta 35, 1776 (1952); NL 6514950; US 3405159 (1966, 1968 both to Merck & Co.). Molecular conformation: Becker et al., Biochem. Biophys. Res. Commun. 41, 444 (1970). Metabolism studies: Shaw et al., J. Biol. Chem. 226, 255 (1957); Calne et al., Br. J. Pharmacol. 37, 57 (1969). Hemodynamic effects in congestive heart failure: S. I. Rajfer et al., N. Engl. J. Med. 310, 1357 (1984). Series of articles on clinical efficacy in Parkinson's disease: Adv. Neurol. 45, 457-510 (1986). Reviews on l-dopa and parkinsonism: Barbeau, Can. Med. Assoc. J. 101, 791 (1969); Pletscher et al., Schweiz. Med. Wochenschr. 100, 797 (1970); Calne, Sandler, Nature 226, 21 (1970); l-Dopa and Parkinsonism, A. Barbeau, Ed. (F. A. Davis, Philadelphia, 1970). Review of acute toxicity data: W. G. Clark et al., Toxicol. Appl. Pharmacol. 28, 1-7 (1974). Comprehensive description: R. Gomez et al., Anal. Profiles Drug Subs. 5, 189-223 (1976).
Properties
Colorless to white, odorless and tasteless crystals or crystalline powder. Needles from water, mp 276-278° (dec) (Yamada); also reported as mp 284-286° (Wysong). [α]D13 13.1° (c = 5.12 in 1N HCl). uv max (0.001N HCl): 220.5, 280 nm (log ε 3.79, 3.42). Readily sol in dil HCl and formic acid. Soly in water: 66 mg/40 ml. Practically insol in ethanol, benzene, chloroform and ethyl acetate. In the presence of moisture, l-dopa is rapidly oxidized by atmospheric oxygen and darkens. LD50 in mice (mg/kg): 3650 ±327 orally, 1140 ±66 i.p., 450 ±42 i.v., >400 s.c.; in male, female rats (mg/kg): >3000, >3000 orally; 624, 663 i.p.; >1500, >1500 s.c. (Clark).Therapeutic Category
Antiparkinsonian.
Keywords
Antiparkinsonian