5472. Levulinic Acid
Nomenclature
CAS number: 123-76-2
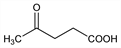
4-Oxopentanoic acid; β-acetylpropionic acid; laevulinic acid; 4-oxovaleric acid. C5H8O3; mol wt 116.12.
C 51.72%, H 6.94%, O 41.33%.
Description and references
Laboratory procedure from starch or cane sugar by boiling with HCl: McKenzie, Org. Synth. coll. vol. I, 335 (1941). Produced commercially from low grade cellulose. By-product of furfural manuf. Extensive review: Leonard, Ind. Eng. Chem. 48, 1331 (1956).
Properties
Plates or leaflets (commercial product is yellow), mp 33-35°. bp 245-246°. d 1.1447. nD16 1.442. Freely sol in water, alcohol, ether; essentially insol in aliphatic hydrocarbons. Protect from light.Derivative
Phenylhydrazone.Nomenclature
CAS number: 588-60-3
Antithermin. C11H14N2O2; mol wt 206.24.
C 64.06%, H 6.84%, N 13.58%, O 15.52%.