Description and references
Oxygenated chalcones isolated from licorice
root, a traditional medicine prepared from various species of Glycyrrhiza (Leguminosae). Exhibit antiparasitic and antitumor
activity and appear to interfere with mitochondrial respiration.
Four have been identified, A-D, as well as a closely related compound, echinatin. Isoln of A and B from G. glabra L. and structure:
T. Saitoh, S. Shibata, Tetrahedron Lett. 50, 4461 (1975); of C and D from G. inflata: K. Kajiyama et al., Phytochemistry 31, 3229 (1992). Mechanism of action study: H. Haraguchi et al., ibid. 48, 125 (1998); L Zhai et al., J.
Antimicrob. Chemother. 43, 793 (1999).
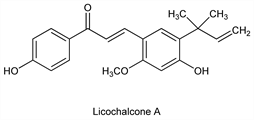
Derivative
Licochalcone A.
Nomenclature
CAS number: 58749-22-7
(2
E)-3-[5-(1,1-Dimethyl-2-propenyl)-4-hydroxy-2-methoxyphenyl]-1-(4-hydroxyphenyl)-2-propen-1-one.
C
21H
22O
4; mol wt 338.40.
C 74.53%, H 6.55%, O 18.91%.
Description and references
Synthesis: A. Islam, M. A. Hossain, Indian J. Chem. 32B, 713 (1993).
HPLC determn in biological fluids: L. Nadelmann et al., J. Chromatogr. B 695, 389 (1997). Antileishmanial activity: M. Chen et al., Antimicrob. Agents Chemother. 38, 1339 (1994). Antimalarial activity: eidem, ibid. 1470. Effect on human cancer cell lines: E. J.
Park et al., Planta Med. 64, 464 (1998).
Properties
Yellow needles from methanol-water, mp 100°. uv
max in methanol: 264, 308, 378 nm (log ε 3.96, 3.97, 4.31).Derivative
Licochalcone B.
Nomenclature
CAS number: 58749-23-8
(2
E)-3-(3,4-Dihydroxy-2-methoxyphenyl)-1-(4-hydroxyphenyl)-2-propen-1-one.
C
16H
14O
5; mol wt 286.28.
C 67.13%, H 4.93%, O 27.94%.
Properties
Yellow needles from methanol-water, mp 197°. uv max in methanol: 262, 366 nm (log ε 3.71, 4.27).Derivative
Licochalcone C.
Nomenclature
CAS number: 144506-14-9
(2
E)-3-[4-Hydroxy-2-methoxy-3-(3-methyl-2-butenyl)phenyl]-1-(4-hydroxyphenyl)-2-propen-1-one.
C
21H
22O
4; mol wt 338.40.
C 74.53%, H 6.55%, O 18.91%.
Properties
Amorphous. uv max in methanol:
250, 308sh, 358 nm (log ε 3.88, 3.95, 4.24).Derivative
Licochalcone D.
Nomenclature
CAS number: 144506-15-0
(2
E)-3-(3,4-Dihydroxy-2-methoxyphenyl)-1-[4-hydroxy-3-(3-methyl-2-butenyl)phenyl]-2-propen-1-one.
C
21H
22O
5; mol wt 354.40.
C 71.17%, H 6.26%, O 22.57%.
Properties
Pale yellow needles from methanol-water, mp 113°. uv max in methanol: 254, 359 nm (log ε 3.86, 4.30).