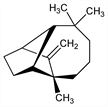
5567. Longifolene
Nomenclature
Description and references
Tricyclic sesquiterpene present to the extent of 5-10% in Indian turpentine oil which is produced commercially from the Himalayan pine, Pinus longifolia Roxb., Pinaceae: J. L. Simonsen, J. Chem. Soc. 117, 570 (1920). Structure: R. H. Moffett, D. Rogers, Chem. Ind. (London) 1953, 916; P. Naffa, G. Ourisson, ibid. 917. Abs config: G. Ourisson, Bull. Soc. Chim. Fr. 1955, 895. Identity of longifolene with junipene and kuromatsuene: S. Akiyoshi et al., Tetrahedron 9, 237 (1960). Total synthesis of naturally occurring (+)-form: E. J. Corey et al., J. Am. Chem. Soc. 86, 478 (1964); of the (±)-form: eidem, ibid. 83, 1251 (1961); J. E. McMurray, S. J. Isser, ibid. 94, 7132 (1972); R. A. Volkmann et al., ibid. 97, 4777 (1975). Review of chemistry of longifolene and its derivs: S. Dev, Acc. Chem. Res. 14, 82-88 (1981); idem, Fortschr. Chem. Org. Naturst. 40, 50-104 (1981).