5574. Loprazolam
Nomenclature
CAS number: 61197-73-7
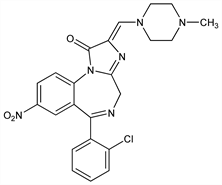
(Z)-6-(2-Chlorophenyl)-2,4-dihydro-2-[(4-methyl-1-piperazinyl)methylene]-8-nitro-1H-imidazo[1,2-a][1,4]benzodiazepin-1-one. C23H21ClN6O3; mol wt 464.90.
C 59.42%, H 4.55%, Cl 7.63%, N 18.08%, O 10.32%.
Description and references
Annelated 1,4-benzodiazepine deriv. Prepn: J. B. Taylor, D. R. Harrison, DE 2605652; eidem, US 4044142 (1976, 1977 both to Roussel-UCLAF). CNS activity: J. R. Agar et al., J. Med. Chem. 20, 1035 (1977). Pharmacological profile: T. G. Johns et al., Arch. Int. Pharmacodyn. Ther. 240, 53 (1979). Effects on sleep in normal volunteers: I. Hindmarch, C. A. Clyde, Drugs Exp. Clin. Res. 2, 61 (1980). Review of pharmacology and efficacy in insomnia: B. G. Clark et al., Drugs 31, 500-516 (1986).
Properties
Crystals from chloroform/ether, mp 214-215°. LD50 in mice: >1000 mg/kg orally (Agar).Derivative
Methanesulfonate.Nomenclature
CAS number: 70111-54-5
Loprazolam mesylate; HR-158; RU-31158; Dormonoct (Hoechst); Havlane (Roussel Diamant); Somnovit (Hosbon); Sonin (Lipha). C23H21ClN6O3.CH3SO3H; mol wt 561.01.
C 51.38%, H 4.49%, Cl 6.32%, N 14.98%, O 17.11%, S 5.72%.
Properties
Crystals from methylene chloride, mp 205-210°.Note
This is a controlled substance (depressant): 21 CFR, 1308.14.Therapeutic Category
Sedative, hypnotic.
Keywords
Sedative/Hypnotic; Benzodiazepine Derivatives