5576. Loracarbef
Nomenclature
Description and references
Carbacephem analog of cefaclor, q.v. Prepn: T. Hirata et al., EP 14476; eidem, US 4708956 (1980, 1987 both to Kyowa Hakko); I. Matsukuma et al., Chem. Pharm. Bull. 37, 1239 (1989). Prepn of the crystalline monohydrate: C. E. Pasini, EP 311366 (1989 to Lilly). Enantioselective synthesis: C. C. Bodurow et al., Tetrahedron Lett. 30, 2321 (1989). Antibacterial spectrum: K. Sato et al., J. Antibiot. 42, 1844 (1989). β-Lactamase stability: R. N. Jones, A. L. Barry, Eur. J. Clin. Microbiol. 6, 570 (1987). Pharmacology in animals: T. Shetler et al., Arzneim.-Forsch. 43, 60 (1993). Pharmacokinetics in children: J. D. Nelson et al., Antimicrob. Agents Chemother. 32, 1738 (1988).
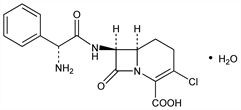
Properties
mp 205-215° (dec) (Matsukuma). [α]D21 +34.0° (c = 0.35 in water).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Carbacephems