5614. Luteolin
Nomenclature
CAS number: 491-70-3
2-(3,4-Dihydroxyphenyl)-5,7-dihydroxy-4H-1-benzopyran-4-one; 3′,4′,5,7-tetrahydroxyflavone; digitoflavone; cyanidenon 1470. C15H10O6; mol wt 286.24.
C 62.94%, H 3.52%, O 33.54%.
Description and references
Found in many plants in glycosidic combination, e.g., as the arabinoside: Perkin, J. Chem. Soc. 69, 800 (1896); Fleischer, Ber. 32, 1186 (1899); Perkin, Horsfall, J. Chem. Soc. 77, 1315 (1900); Hayashi, Inoue, Acta Phytochim. 15, 53 (1949); C.A. 43, 8450 (1949). Identity with digitoflavone: Kiliani, Mayer, Ber. 34, 3577 (1901). Synthesis: Hutchins, Wheeler, J. Chem. Soc. 1939, 91. See also Bioflavonoids.
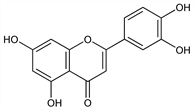
Derivative
Monohydrate.Properties
Yellow needles from alc, dec 328-330°. Sublimes in high vacuum. Sparingly sol in water. Sol in alkalies forming yellow solns.Derivative
5-Glucoside.Nomenclature
Galuteolin.C21H20O11; mol wt 448.38.
C 56.25%, H 4.50%, O 39.25%.
Properties
From seeds of Galega officinalis L., Leguminosae: Barger, White, Biochem. J. 17, 836 (1923). Structure: Nakamura, Hukuti, J. Pharm. Soc. Jpn. 60, 449 (1940); C.A. 34, 79103 (1940); Nordstrm, Swain, J. Chem. Soc. 1953, 2764. Yellow needles from hot dil alc, dec 280°. Practically insol in water. Slightly sol in abs alcohol; sol in hot dil alcohol.Derivative
7-Glucoside.Nomenclature
Cynaroside.C21H20O11; mol wt 448.38.
C 56.25%, H 4.50%, O 39.25%.