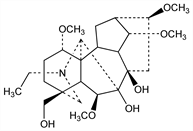
5618. Lycoctonine
Nomenclature
CAS number: 26000-17-9
(1α,6β,14α,16β)-20-Ethyl-4-(hydroxymethyl)-1,6,14,16-tetramethoxyaconitane-7,8-diol; royline. C25H41NO7; mol wt 467.60.
C 64.21%, H 8.84%, N 3.00%, O 23.95%.
Description and references
Isomeric with delsoline. Originally isolated from Aconitum lycoctonum L., Ranunculaceae: Hubschmann, Schweiz. Wochschr. Pharm. 3, 269 (1865). Widely distributed in Aconitum and Delphinium spp., Ranunculaceae. Structure: Edwards et al., Can. J. Chem. 34, 1315 (1956); Anet et al., ibid. 35, 400 (1957). Identity with royline: Edwards, Rodger, ibid. 37, 1187 (1959). Stereochemistry: Przybylska, Marion, ibid. 1843. Revised configuration: S. W. Pelletier et al., J. Am. Chem. Soc. 103, 6536 (1981).
Properties
Crystals. Bitter taste. mp 143°. [α]D20 +53° (ethanol). Slightly sol in water, ether, benzene; freely sol in alcohol, chloroform.Derivative
Ajacine.Nomenclature
CAS number: 509-17-1
N-Acetylanthranilic acid ester. C34H48N2O9; mol wt 628.75.
C 64.95%, H 7.69%, N 4.46%, O 22.90%.
Properties
Needles from 70% alcohol, mp 154°. [α]D22 +49.5° (c = 2 in abs alcohol); [α]D16 +53° (c = 0.66 in chloroform). uv max: 223, 252, 310 nm (ε 28400, 16600, 5400). Slightly sol in water; sol in ether, alcohol with blue fluorescence.Derivative
Lycaconitine.Nomenclature
CAS number: 25867-19-0
N-Succinylanthranilic acid ester. C36H48N2O10; mol wt 668.77.
C 64.65%, H 7.23%, N 4.19%, O 23.92%.
Description and references
Isoln: Schulze, Bierling, Arch. Pharm. 251, 8 (1913).