5645. Macromerine
Nomenclature
CAS number: 2970-95-8
α-[(Dimethylamino)methyl]-3,4-dimethoxybenzenemethanol; α-[(dimethylamino)methyl]veratryl alcohol; 3,4-dimethoxy-α-[(dimethylamino)methyl]benzyl alcohol; dimethylaminomethyl 3,4-dimethoxyphenyl carbinol. C12H19NO3; mol wt 225.28.
C 63.98%, H 8.50%, N 6.22%, O 21.31%.
Description and references
Prepn of dl-form: La Manna et al., Farmaco Ed. Sci. 15, 9 (1960); Chapman et al., Proc. R. Soc. London Ser. B 163, (990), 116 (1965); Hodgkins et al., Tetrahedron Lett. 1967, 1321; of d- and l-forms: La Manna et al., loc. cit. Isoln of l-form from cactus, Coryphantha macromeris (Engelm.) Lem., Britton and Rose, Cactaceae: Hodgkins et al., loc. cit.
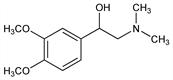
Derivative
dl-Form.Properties
Crystals, mp 46-47°.Derivative
l-Form.Properties
Crystals, mp 66-67.5°. [α]D25 -147.01° (c = 0.0390 g/ml chloroform), -42.61° (c = 0.0200 g/ml abs alcohol).Derivative
d-Form.Properties
Crystals, mp 60-62°.Derivative
dl-Form hydrochloride.C12H19NO3.HCl; mol wt 261.75.
C 55.06%, H 7.70%, N 5.35%, O 18.34%, Cl 13.54%.