5648. Maduramicin
Nomenclature
CAS number: 84878-61-5
(2R,3S,4S,5R,6S)-6-[(1R)-1-[(2S,5R,7S,8R,9S)-2-[(2S,2′R,3′S,5R,5′R)-3′-[(2,6-Dideoxy-3,4-di-O-methyl-β-l-arabino-hexopyranosyl)oxy]octahydro-2-methyl-5′-[(2S,3S,5R,6S)-tetrahydro-6-hydroxy-3,5,6-trimethyl-2H-pyran-2-yl][2,2′-bifuran]-5-yl]-9-hydroxy-2,8-dimethyl-1,6-dioxaspiro[4.5]dec-7-yl]ethyl]tetrahydro-2-hydroxy-4,5-dimethoxy-3-methyl-2H-pyran-2-acetic acid, monoammonium salt; antibiotic X-14868A ammonium salt; CL-273703; Cygro (Am. Cyanamid). C47H83NO17; mol wt 934.16.
C 60.43%, H 8.96%, N 1.50%, O 29.12%.
Description and references
Polyether antibiotic chemically related to the lonomycins, q.v. Isoln as sodium salt from Nocardia sp X-14868 and biological activity: C.-M. Liu et al., US 4278663 (1981 to Hoffmann-La Roche); as free acid hydrate from Actinomadura yumaense sp nov.: D. P. Labeda et al., US 4407946 (1983 to Am. Cyanamid). Fermentation and properties: C.-M. Liu et al., J. Antibiot. 36, 343 (1983). 13C-NMR spectrum: S. Rajan, ibid. 37, 1495 (1984). Biosynthetic studies: H.-R. Tsou et al., ibid. 1651; H.-R. Tsou et al., ibid. 40, 94 (1987). Antimalarial activity: L. Oronsky, US 4496549 (1985 to Am. Cyanamid). Nematocidal activity: I. B. Wood, US 4510134 (1985 to Am. Cyanamid).
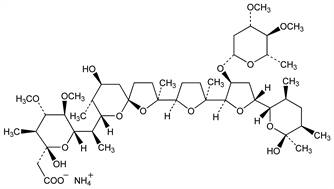
Derivative
Sodium salt.C47H79NaO17; mol wt 939.11.
C 60.11%, H 8.48%, Na 2.45%, O 28.96%.