Nomenclature
CAS number: 110-16-7
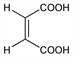
(
Z)-Butenedioic acid; toxilic acid;
cis-1,2-ethylenedicarboxylic acid.
C
4H
4O
4; mol wt 116.07.
C 41.39%, H 3.47%, O 55.14%.
Description and references
Prepd by the catalytic oxidation of benzene
over vanadium pentoxide: Bhattacharyya, Venkataraman, J. Appl. Chem. 8, 728 (1958);
Saffer, Olenberg, FR 1321416 (1963 to Scientific Design), C.A. 59, 11265d (1963). Crystal structure:
Shahat, Acta Crystallogr. 5, 763 (1952). Review: W. D. Robinson, R. A. Mount
in Kirk-Othmer Encyclopedia of Chemical Technology vol. 14 (Wiley-Interscience, New York, 3rd ed., 1981)
pp 770-793.
Properties
White crystals from water, mp 138-139°; from alcohol and benzene, mp 130-131°. Faint,
acidulous odor; characteristic repulsive, astringent taste. d 1.59. Is converted in
part into the much higher-melting fumaric acid (mp 287°) when heated
to a temp slightly above the melting point. Freely sol in water or
alcohol. Sol in acetone, glacial acetic acid. Slightly sol in ether.
Practically insol in benzene.Caution
Strong irritant.Use
Manuf artificial resins; to retard rancidity of
fats and oils in 1:10,000 (these are said to keep 3 times longer than
those without the acid); dyeing and finishing wool, cotton, and silk;
preparing the maleate salts of antihistamines and similar drugs.
