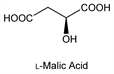
5707. Malic Acid
Nomenclature
Description and references
The naturally occurring isomer is the l-form which has been found in apples and many other fruits and plants. Prepn of d- and dl-forms, and resolution of racemic mixture: McKenzie et al., J. Chem. Soc. 123, 2875 (1923). Solubilities: Descamps, Bull. Soc. Chim. Belg. 49, 91 (1940). Microbial production of l-form: Kitahara; Abe et al., US 2972566; US 3063910 (1961, 1962, both to Kyowa). Configuration: J. A. Mills, W. Klyne in Progress in Stereochemistry vol. 1, W. Klyne, Ed. (Academic Press, New York, 1954) pp 182-183; E. L. Eliel, Stereochemistry of Carbon Compounds (McGraw-Hill, New York, 1962) pp 97-98; Cymerman-Craig, Roy, Tetrahedron 21, 1847 (1965). Review of uses: Manuf. Chem. Aerosol News 35, 56 (December, 1964). Review: S. E. Berger, “Hydroxy Dicarboxylic Acids” in Kirk-Othmer Encyclopedia of Chemical Technology vol 13 (Wiley-Interscience, New York, 3rd ed., 1981) pp 103-110.