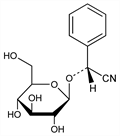
5720. Mandelonitrile Glucoside
Nomenclature
Description and references
Prepn of d-form by action of yeast on amygdalin: Fischer, Ber. 28, 1508 (1895); Auld, J. Chem. Soc. 93, 1276 (1908). Isoln of d-form from Prunus serotina, Ehrk, P. macrophylla Sieb et Zucc., Rosaceae: Power, Moore, ibid. 95, 243 (1909); 97, 1099 (1910); Kariyone, Matsushima, J. Pharm. Soc. Jpn. No. 514, 1061 (1924); from Eucalyptus corynocalyx F.v.M., Myrtaceae: Finnemore et al., J. Proc. R. Soc. N.S.W. 69, 209 (1936); from Pteridium aquilinum (L.) Kühn, Polypodiaceae: Kofod, Eyjolfssen, Tetrahedron Lett. 1966, 1289. Isoln of dl-form from Prunus laurocerasus L. and Cotoneaster microphylla Wall., Rosaceae: Winkler, Simon, Ann. 31, 263 (1839); Hérissey, J. Pharm. Chim. 24, 537 (1906). Isoln of l-form from Sambucus nigra L., Caprifoliaceae: Guignart, Compt. Rend. 141, 16 (1905); Bourquelot, Danjot, ibid. 598 (1905). Prulaurasin is dl-mandelonitrile glucoside and sambunigrin is l-mandelonitrile glucoside: Caldwell, Courtauld, J. Chem. Soc. 91, 671 (1907). Biosynthesis of d-mandelonitrile glucoside: Mentzer, Favre-Bonvin, Compt. Rend. 253, 1072 (1961). Abs config of isomers: U. Schwarzmaier, Ber. 109, 3250 (1976).