5743. Manidipine
Nomenclature
CAS number: 89226-50-6
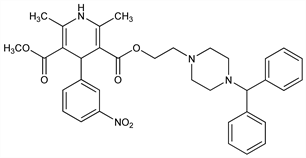
1,4-Dihydro-2,6-dimethyl-4-(3-nitrophenyl)-3,5-pyridinedicarboxylic
acid 2-[4-(diphenylmethyl)-1-piperazinyl]ethyl methyl ester; 2-[4-(diphenylmethyl)-1-piperazinyl]ethyl methyl (±)-1,4-dihydro-2,6-dimethyl-4-(m-nitrophenyl)-3,5-pyridinecarboxylate; franidipine. C35H38N4O6; mol wt 610.70.
C 68.83%, H 6.27%, N 9.17%, O 15.72%.
Description and references
Dihydropyridine calcium channel blocker. Prepn: K. Meguro, A. Nagaoka, EP 94159; eidem, US 4892875 (1983, 1990, both to Takeda); K. Meguro et al., Chem. Pharm. Bull. 33, 3787 (1985). Synthesis and bioactivity of optical isomers: M. Kajino et al., ibid. 37, 2225 (1989). Acute toxicity study: S. Chiba et al., Yakuri to Chiryo 17, Suppl. 4, 961 (1989), C.A. 111, 146499b (1989). Determn of enantiomers in human serum: M. Yamaguchi et al., J. Chromatogr. 575, 123 (1992). Clinical evaluation in hypertension: K. Mizuno et al., Curr. Ther. Res. 52, 248 (1992).
Properties
Light yellow crystals from isopropyl ether + hexane, mp 125-128°.Derivative
Dihydrochloride.Nomenclature
CAS number: 89226-75-5
CV-4093; Calslot (Takeda). C35H38N4O6.2HCl; mol wt 683.62.
C 61.49%, H 5.90%, N 8.20%, O 14.04%, Cl 10.37%.
Properties
Two crystalline forms. α-form: yellow crystals, mp 157-163°. β-form: light yellow, fine crystals, mp 174-180°. Also prepd as the monohydrate, mp 167-170°. LD50 in male, female mice, male, female rats (mg/kg): 387, 340, 222, 199 s.c.; 62.2, 68.0, 66.5, 48.8 i.p.; 190, 171, 247, 156 orally (Chiba).Therapeutic Category
Antihypertensive.
Keywords
Antihypertensive; Dihydropyridine Derivatives; Calcium Channel Blocker; Dihydropyridine Derivatives