5758. Matricarin
Nomenclature
CAS number: 5989-43-5
(3S,3aR,4S,9aS,9bR)-4-(Acetyloxy)-3,3a,4,5,9a,9b-hexahydro-3,6,9-trimethylazuleno[4,5-b]furan-2,7-dione. C17H20O5; mol wt 304.34.
C 67.09%, H 6.62%, O 26.29%.
Description and references
Isoln from Matricaria chamomilla L. and Artemisia tilesii Ledeb, Compositae: Z. Cekan et al., Collect. Czech. Chem. Commun. 24, 1554 (1959). Structure: W. Herz, K. Ueda, J. Am. Chem. Soc. 83, 1139 (1961). Molecular conformation: M. Martinez V. et al., J. Nat. Prod. 51, 221 (1988).
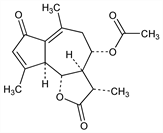
Properties
Plates from benzene + petr ether and acetone + petr ether, mp 190-191°. [α]D23 +23.5° (c = 0.65 in chloroform). uv max: 255 nm (ε 15,100).Derivative
Desacetoxymatricarin.Nomenclature
CAS number: 17946-87-1
C15H18O3; mol wt 246.30.
C 73.15%, H 7.37%, O 19.49%.
Description and references
Isoln from Artemisia spp.: M. Holub, V. Herout, Collect. Czech. Chem. Commun. 27, 2980 (1962); H. H. A. Linde, M. S. Ragab, Helv. Chim. Acta 50, 1961 (1967). Synthesis and stereochemistry: E. H. White et al., Tetrahedron 25, 2099 (1969).
Properties
Crystals from ether, mp 205-205.5°, also reported as mp 202-203° (Holub). [α]D24 +52.5° (c = 1 in chloroform).Derivative
Desacetylmatricarin monohydrate.C15H18O4.H2O; mol wt 280.32.
C 64.27%, H 7.19%, O 28.54%.
Properties
Crystals from benzene + acetone, mp 123-125°, resolidifies and mp 143-146°.Derivative
Tetrahydromatricarin.C17H24O5; mol wt 308.37.
C 66.21%, H 7.84%, O 25.94%.