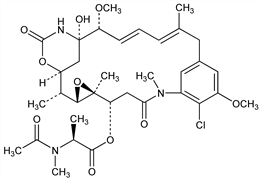
5761. Maytansine
Nomenclature
Description and references
Anti-leukemic ansa macrolide, member of a class of compounds that includes the rifamycins, streptovaricins etc. First ansa compound isolated from a plant rather than produced by a microorganism. Isoln from Maytenus ovatus Loes., Celastraceae and structure elucidation: S. M. Kupchan et al., J. Am. Chem. Soc. 94, 1354 (1972); S. M. Kupchan, DE 2241418 (1974); S. M. Kupchan et al., US 3896111 (1975 to Research Corp.). Crystal structure and absolute configuration of the (3-bromopropyl) ether: R. F. Bryan et al., J. Chem. Soc. Perkin Trans. 2 1973, 897. Synthetic studies: A. I. Meyers et al., Tetrahedron Lett. 1974, 717; 1975, 1745, 1749; E. J. Corey, M. G. Bock, ibid. 1975, 2643; E. J. Corey et al., ibid. 1978, 1051; A. I. Meyers, J. P. Hudspeth, ibid. 22, 3925 (1981). Total synthesis of (±)-form: A. I. Meyers et al., J. Am. Chem. Soc. 102, 6597 (1980); of naturally occurring (-)-form: E. J. Corey et al., ibid. 6613. Cytotoxic action: M. K. Wolpert-Defilippes, Biochem. Pharmacol. 24, 751 (1975). Clinical studies in carcinoma, melanoma: J. A. Neidhart et al., Cancer Treat. Rep. 64, 675 (1980); D. L. Ahmann et al., ibid. 721. Toxicity: G. M. Mugera, J. M. Ward, ibid. 61, 1333 (1977).