5805. Megestrol Acetate
Nomenclature
Description and references
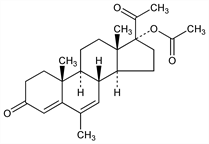
Orally active progestogen; formerly used in combinations as oral contraceptive. Prepn: Ringold et al., J. Am. Chem. Soc. 81, 3712 (1959); Dodson, Sollman, US 2891079 (1959 to Searle); Kirk et al., GB 870286 C.A. 56, 10248i (1962), and US 3356573 (1967 to Brit. Drug Houses); Cross, US 3400137 (1968 to Syntex). Biological effectiveness studies: Chang, Kincl, Steroids 12, 689 (1968). Metabolic studies: Cooper, Kellie, ibid. 11, 133 (1968). Soly and diffusion studies: Sundaram, Kincl, ibid. 12, 517 (1968). Comparative clinical trial with tamoxifen in metastatic breast cancer: J. C. Allergra et al., Semin. Oncol. 12, Suppl. 6, 61 (1985). Evaluation in prevention of hot flashes: C. L. Loprinzi et al., N. Engl. J. Med. 331, 347 (1994). Review of carcinogenicity studies: IARC Monographs 21, 431-439 (1979).
Properties
Crystals from methanol, mp 214-216°. Soly at 37°: in water (2 μg/ml); in plasma (24 μg/ml). [α]D24 +5° (chloroform). uv max (ethanol): 287 nm (log ε 4.40).Derivative
Mixture with ethinyl estradiol.Nomenclature
Derivative
Mixture with mestranol.Nomenclature
Therapeutic Category
Progestogen. Palliative treatment of breast and endometrial carcinoma.
Therapeutic Category (Veterinary)
Progestogen. Estrus regulator.Keywords
Antineoplastic (Hormonal); Progestogens; Progestogen